Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Sugikawa, Susumu; Umeda, Miki; Kobayashi, Fuyumi; Nagata, Masanobu*; Dojiri, Shigeru; Amano, Masae*
Proceedings of International Conference on Nuclear Energy System for Future Generation and Global Sustainability (GLOBAL 2005) (CD-ROM), 4 Pages, 2005/10
The mineralization of radioactive contaminated organic wastes by mediated electrochemical oxidation process has some attractive features as alternative to incineration process: The process operates safely at low temperatures and ambient pressures. JAERI has been investigated the process since 1996 and confirmed complete mineralization of this organic solvent. In order to greatly improve current efficiency for the oxidation reaction, further experiments were performed under condition of strong mixing of organic solvent and anolyte with an aide of ultrasonic wave. The current efficiencies for the oxidation reaction by ultrasonic agitation between organic solvent and anolyte were twice to that by mechanical agitation. On the basis of these results, two processes, one for destruction of a small amount of TBP/dodecane and the other for destruction of intermediate compounds following alkaline hydrolysis of a large amount of TBP/dodecane, were proposed.
 -tetraoctyl-3-oxapentanediamide (TODGA) from nitric acid into
-tetraoctyl-3-oxapentanediamide (TODGA) from nitric acid into  -dodecane
-dodecaneZhu, Z.-X.; Sasaki, Yuji; Suzuki, Hideya*; Suzuki, Shinichi; Kimura, Takaumi
Analytica Chimica Acta, 527(2), p.163 - 168, 2004/12
Times Cited Count:267 Percentile:99.09(Chemistry, Analytical)The extraction of 75 elements by  -tetraoctyl-3-oxapentanediamide (TODGA) from nitric acid to
-tetraoctyl-3-oxapentanediamide (TODGA) from nitric acid to  -dodecane was investigated and discussed concerning distribution ratios (D) of metal ions and their ionic radii. The extractability for elements depends strongly on their oxidation state. Monovalent and pentavalent ions were not extractable. In divalent metal extraction, Ca(II) (ionic radius: 100 pm) showed the highest D value and the D decreased with increase or decrease of the ionic radius. Tri- and tetravalent ions with ionic radii of 87-113 pm and 83-94 pm, respectively, showed the D over 1000, and the extracted chemical forms were determined to be M(TODGA)
-dodecane was investigated and discussed concerning distribution ratios (D) of metal ions and their ionic radii. The extractability for elements depends strongly on their oxidation state. Monovalent and pentavalent ions were not extractable. In divalent metal extraction, Ca(II) (ionic radius: 100 pm) showed the highest D value and the D decreased with increase or decrease of the ionic radius. Tri- and tetravalent ions with ionic radii of 87-113 pm and 83-94 pm, respectively, showed the D over 1000, and the extracted chemical forms were determined to be M(TODGA) (NO
(NO )
)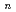 or M(TODGA)
or M(TODGA) (NO
(NO )
)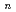 (
( =3 or 4). The oxonium ions with higher oxidation states than tetravalent one forms the complicated metal-complex with TODGA, and the number of TODGA associated in those extraction reactions was confirmed to be 2 at most.
=3 or 4). The oxonium ions with higher oxidation states than tetravalent one forms the complicated metal-complex with TODGA, and the number of TODGA associated in those extraction reactions was confirmed to be 2 at most.
Uchiyama, Gunzo; Hotoku, Shinobu; Fujine, Sachio
Solvent Extr. Ion Exch., 16(5), p.1177 - 1190, 1998/00
Times Cited Count:9 Percentile:48.11(Chemistry, Multidisciplinary)no abstracts in English
*; *; *; Naito, Yoshitaka; Kurosawa, Masayoshi
JAERI-Research 95-033, 31 Pages, 1995/03
no abstracts in English
Fujine, Sachio; Uchiyama, Gunzo; Maeda, Mitsuru; R.Thompson*; C.Mason*; R.Bush*
Solvent Extraction in the Process Industries, Vol. 3, p.1789 - 1796, 1993/00
no abstracts in English
Journal of Nuclear Science and Technology, 4(8), p.427 - 430, 1967/00
Times Cited Count:2no abstracts in English